Coordination Compounds
What is crystal field splitting energy? How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
The degenerate d-orbitals (in a spherical field environment) split into two levels i.e., eg and t2g in the presence of ligands. The splitting of the degenerate levels due to the presence of ligands is called the crystal-field splitting while the energy difference between the two levels (eg and t2g) is called the crystal-field splitting energy. It is denoted by ∆o.
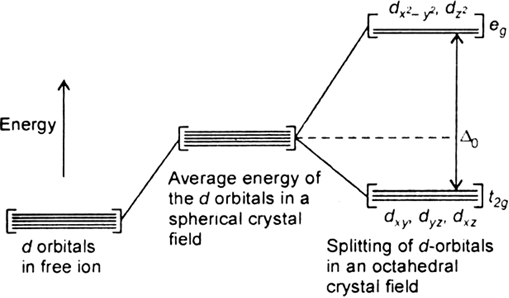
Fig. d-orbital splitting in an octahedral crystal field.
The formation of complex depend on the crystal field splitting, ∆o and pairing energy (P).
i)If ∆o < P, the fourth electron enters one of the eg orbitals giving theconfiguration t2g3. Ligands for which ∆o < P are known as weak field ligands and form high spin complexes.
ii) If ∆o > P, it becomes more energetically favourable for the fourth electron to occupy a t2g orbital with configuration t2g4 eg0. Ligands which produce this effect are known as strong field ligands and form low spin complexes.
Sponsor Area
Some More Questions From Coordination Compounds Chapter
Indicate the type of isomerism exhibited by the following complexes and draw structures for these isomers:
(i) K[Cr(H2O)2(C2O4)2], (ii) [Co(en)3Cl3,
(iii) [Co(NH3)5(NO2)]|NO3]2, (iv) [Pt(NH3)(H2O)Cl2]
Explain on the basis of valence bond theory that [Ni(CN)4]2– ion with square planar is diamagnetic and the [NiCl4]2– ion with tetrahedral geometry is paramagnetic.
Predict the number of unpaired electrons in the square planar [Pt(CN)4,]2– ion.
The hexaquo manganese(II) ion contains five unpaired electrons, while the hexacynoion contains only one unpaired electron. Explain using crystal field theory.
Calculate the overall complex dissociation equilibrium constant for the Cu(NH3)42+ion, given β4 for this complex is 2.1 x 1013.
Identify the ligands from the following coordination compounds:
(a) [Co(en)2Cl(NO2)2]
(b) K[Co(CN)(CO2)(NO)]
(c) K3[Al(OH)6]
(d) [Co(H2O)2(NH3)4](OH)3
Mock Test Series
Sponsor Area
NCERT Book Store
NCERT Sample Papers
Sponsor Area