Haloalkanes and Haloarenes
(a) Draw the structures of major monohalo products in each of the following reactions :
(b) Which halogen compound in each of the following pairs will react faster in SN2 reaction:
(i) CH3Br or CH3I
(ii) (CH3)3C−Cl or CH3−Cl
a)
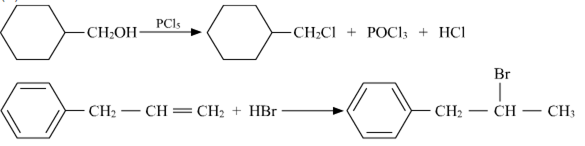
b)
(i) CH3I will react faster in SN2 reaction than CH3Br. This is because I− is a better leaving group, owing to its greater size than Br−. As a result, it will leave at a faster rate in the presence of an incoming nucleophile.
(ii) CH3−Cl will react faster in SN2 reaction than (CH3)3 C−Cl, as CH3−Cl is a primary halide whereas (CH3)3C−Cl is a tertiary halide. Primary halides undergo SN2 reactions faster.
Sponsor Area
Some More Questions From Haloalkanes and Haloarenes Chapter
Why is sulphuric acid not used during the reaction of alcohols with KI?
Write structures of different dihalogen derivatives of propane.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
i) A single monochloride.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
i)Three isomeric monochlorides.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
Four isomeric monochlorides.
Draw the structures of major monohalo products in each of the following reactions:![]()
Draw the structures of major monohalo products in each of the following reactions: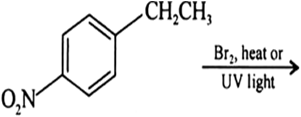
Draw the structures of major monohalo products in each of the following reactions: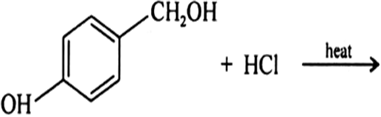
Draw the structures of major monohalo products in each of the following reactions: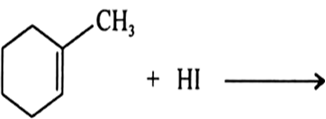
Mock Test Series
Sponsor Area
NCERT Book Store
NCERT Sample Papers
Sponsor Area