Haloalkanes and Haloarenes
Which would undergo SN1 reaction faster in the following pair?

A tertiary alkyl halide tends to undergo the SN1 mechanism because it can form a tertiary carbocation, which is stabilized by the three alkyl groups attached to it. As alkyl groups are electron donating, they allow the positive charge in the carbocation to be delocalized by the induction effect. Hence, out of the given pairs, (CH3)3C-Br would undergo SN1 reaction faster than CH3-CH2-Br.
Order of stability: tertiary>secondary>primary
Sponsor Area
Some More Questions From Haloalkanes and Haloarenes Chapter
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
Four isomeric monochlorides.
Draw the structures of major monohalo products in each of the following reactions:![]()
Draw the structures of major monohalo products in each of the following reactions: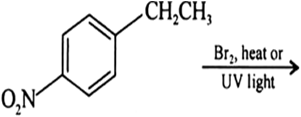
Draw the structures of major monohalo products in each of the following reactions: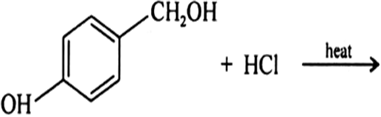
Draw the structures of major monohalo products in each of the following reactions: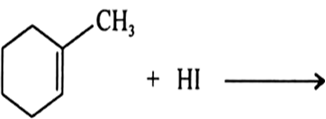
Draw the structures of major monohalo products in each of the following reactions: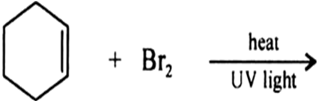
Arrange each set of compounds in order of increasing boiling points:
Bromomethane, Bromoform, Chloro-methane, Dibromomethane.
Arrange each set of compounds in order of increasing boiling points:
1-chloropropane, Isopropyl chloride, 1-chlorobutane.
Arrange each set of compounds in order of increasing boiling points:![]()
Mock Test Series
Sponsor Area
NCERT Book Store
NCERT Sample Papers
Sponsor Area