Haloalkanes and Haloarenes
Why (-NO2) group shows its effect only at ortho- and para-position and not at meta–position?
Presence of nitro group at ortho- and para-positions withdraws the electron density from the benzene ring and thus facilitates the attack of the nucleophile on haloarene. The carbanion thus formed is stabilised through resonance. The negative charge appeared at ortho- and para- positions with respect to the halogen substituent is stabilised by –NO2 group.
Sponsor Area
Some More Questions From Haloalkanes and Haloarenes Chapter
Write structures of the following compounds:
1, 4-Dibromo but-2-ene
Write structures of the following compounds:
1-Bromo-4-sec butyl-2-methyl benzene.
Why is sulphuric acid not used during the reaction of alcohols with KI?
Write structures of different dihalogen derivatives of propane.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
i) A single monochloride.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
i)Three isomeric monochlorides.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields:
Four isomeric monochlorides.
Draw the structures of major monohalo products in each of the following reactions:![]()
Draw the structures of major monohalo products in each of the following reactions: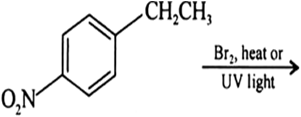
Draw the structures of major monohalo products in each of the following reactions: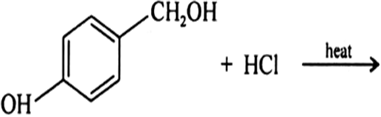
Mock Test Series
Sponsor Area
NCERT Book Store
NCERT Sample Papers
Sponsor Area