Sponsor Area
Coordination Compounds
(i) Write the IUPAC name of the complex [Cr(NH3)4Cl2]Cl.
(ii) What type of isomerism is exhibited by the complex [Co(en)3]3+?
(en = ethane-1,2-diamine)
(iii) Why is [NiCl4]2− paramagnetic but [Ni(CO)4] is diamagnetic?
(At. nos. : Cr = 24, Co = 27, Ni = 28)
(i) The IUPAC name of the complex [Cr(NH3)4Cl2]Cl is Tetraamminedichlorochromium(III) chloride.
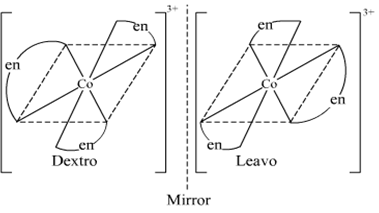
(ii) The complex [Co(en)3]3+ exhibits optical isomerism. Its optical isomers are shown below.
(iii) In [NiCl4]2−, the oxidation state of Ni is +2. Chloride is a weak field ligand and does not cause pairing up of electrons against the Hund's rule of maximum multiplicity. As a result, two unpaired electrons are present in the valence d-orbitals of Ni which impart paramagnetic character to the complex. On the other hand, carbonyl is a strong field ligand and causes pairing up of electrons against the Hund's rule of maximum multiplicity. As a result, no unpaired electrons are present and hence, the complex is diamagnetic.
Some More Questions From Coordination Compounds Chapter
Write the formula for the following coordination compound:
Dichloridobis(ethane-1, 2-diamine)platinum(lV) nitrate.
Write the formula for the following coordination compound:
Iron(III) hexacyanidoferrate(II)
Write the IUPAC names of the following coordination compounds:
(i) [Co(NH3)6]Cl3
(ii) [Co(NH3)5Cl]Cl2
(iii) K3[Fe(CN)6]
(iv) K3[Fe(C2O4)3]
(v) K2[PdCl4]
(vi) [Pt(NH3)2Cl(NH2CH3)]Cl.
Indicate the type of isomerism exhibited by the following complexes and draw structures for these isomers:
(i) K[Cr(H2O)2(C2O4)2], (ii) [Co(en)3Cl3,
(iii) [Co(NH3)5(NO2)]|NO3]2, (iv) [Pt(NH3)(H2O)Cl2]
(i) K[Cr(H2O)2(C2O4)2], (ii) [Co(en)3Cl3,
(iii) [Co(NH3)5(NO2)]|NO3]2, (iv) [Pt(NH3)(H2O)Cl2]
Explain on the basis of valence bond theory that [Ni(CN)4]2– ion with square planar is diamagnetic and the [NiCl4]2– ion with tetrahedral geometry is paramagnetic.
Predict the number of unpaired electrons in the square planar [Pt(CN)4,]2– ion.
Sponsor Area
Mock Test Series
Mock Test Series





