Discuss the use of separating funnels.
Separating funnels are used to separate two liquids which do not form homogeneous mixture on mixing but form two distinct layers. The principle is that immiscible liquids separate out in layers depending on their densities. The immiscible liquids like water and mustard oil when mixed form two separate layers, hence they can be separated using separating funnels.
Experiment. Take the mixture of water and mustard oil in a separating funnel. Allow the mixture to stand undisturbed for sometime. When separate layers of oil and water are formed, oil being lighter makes the upper layer. Open the stopcock of the separating funnel and pour out the lower layer of water into the beaker carefully. Oil remaining behind is then collected in another beaker or flask.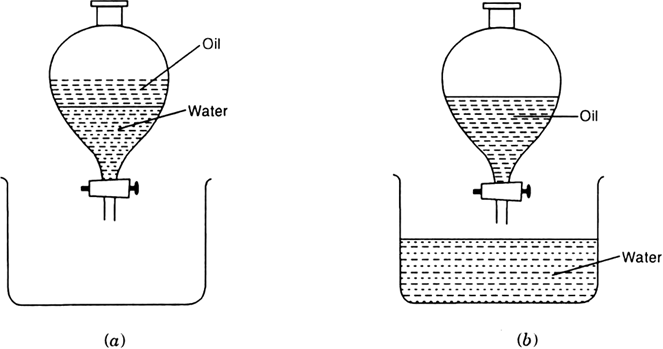
Fig. (a) Mixture (b) pure liquids.



