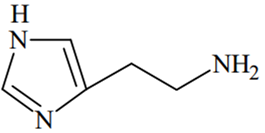
The predominant form of histamine present in human blood is (pKa, Histidine = 6.0)
A.
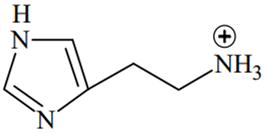
The N-atoms present in the ring will have the same pKa values (6.0), while N atom outside the ring will have different pKa value (pKa > 7.4)
Therefore, two N-atoms inside the ring will remain in the unprotonated form in human blood because of their pKa(6.0) < pH of blood (7.4), while the N-atom outside the ring will remain in protonated form because of its pKa > pH of blood (7.4).