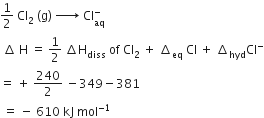Question
Oxidising power of chlorine in aqueous solution can be determined by the parameters indicated below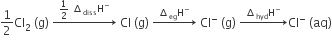
The energy involved in conversion of 
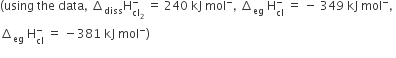
-
152 kJ mol-
-
-610 kJ mol-
-
-850 kJ mol-
-
+120 kJ mol-
Solution
B.
-610 kJ mol-