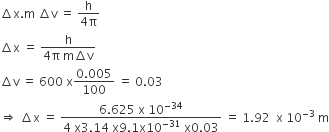In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainty with which the position of the electron can be located is ( h = 6.6 x10-34 kgm2s-1 mass of electron, em= 9.1 x10-31kg)
-
1.52 x 10-4m
-
5.10 x 10-3m
-
1.92 x 10-3m
-
3.84 x 10-3m
C.
1.92 x 10-3m