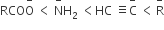Question
The correct order of increasing basicity of the given conjugate bases (R = CH3) is
Solution
A.

Stronger acid has a weaker conjugate base.
Order of acidity : RCOOH > CH≡CH > NH3 > RH
Order of basicity :