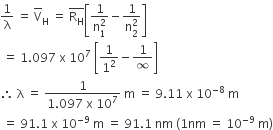Which of the following sets of quantum numbers is correct for an electron in 4f orbital?
-
n = 4, I =3, m = +4, s = + 1/ 2
-
n = 3, I = 2, m = -2, S = + 1/2
-
n =4, I = 3, m = +1, s = + 1/ 2
-
n =4, I = 4, m -4, s = -1/ 2
C.
n =4, I = 3, m = +1, s = + 1/ 2
For 4f orbital electrons, n = 4
s p d f
l = 3 (because 0 1 2 3 )
m = +3, +2, +1, 0, –1, –2, –3
s = ±1/2.